
If CP and CV denote the specific heats of nitrogen per unit mass at constant pressure and constant volume respectively, then.

OpenFOAM-9 multiphaseEulerFoam, wallBoiling, changing table Hs to accommodate new pressure : r/OpenFOAM

Energy Conversion CHE 450/550. Ideal Gas Basics and Heat Capacities - I Ideal gas: – a theoretical gas composed of a set of non-interacting point particles. - ppt download


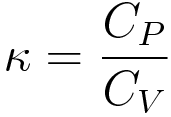





![Interpretation of kappa according to Landis and Kosh [22] | Download Table Interpretation of kappa according to Landis and Kosh [22] | Download Table](https://www.researchgate.net/publication/267933214/figure/tbl1/AS:669227782524962@1536567745506/nterpretation-of-kappa-according-to-Landis-and-Kosh-22.png)






